
Every tablet, capsule, or syrup that reaches a patient is the result of hundreds of controlled activities, strict documentation, and continuous quality checks. Unlike other manufacturing sectors, pharmaceutical production operates in a highly regulated environment where precision, traceability, and compliance are critical.
A minor deviation in formulation, documentation, or handling can lead to regulatory action, product recalls, financial losses, customer defamation and reputational damage. In this context, the way a pharma manufacturer manages operations is not merely an operational decision but a strategy to grow. This is why choosing the right Enterprise Resource Planning (ERP) system is fundamental to long-term success.
Challenges Encountered With Manual Systems:
Despite strict regulatory requirements in Philippines and Southeast Asia, many pharmaceutical companies still relied on spreadsheets, registers, and basic software tools. While these methods may work at a small scale, they become difficult to manage as operations grow.
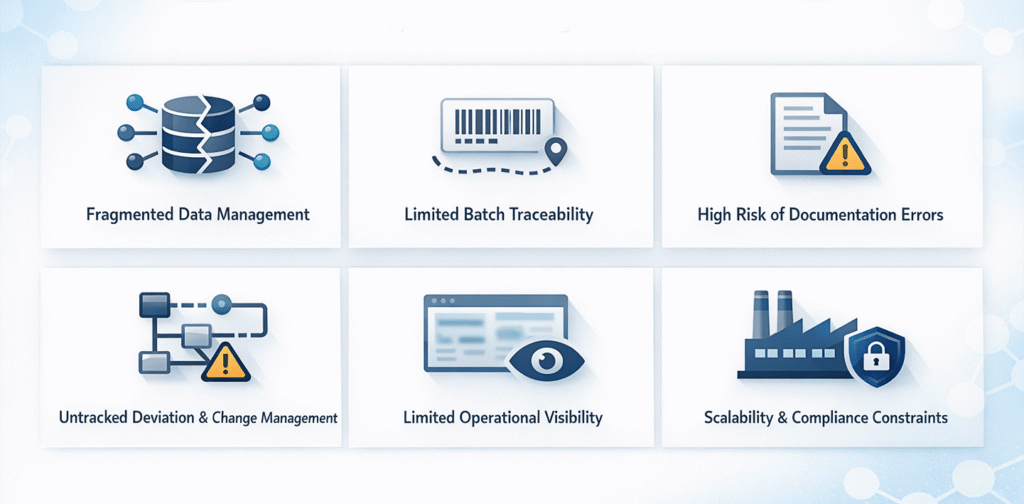
Fragmented Data Management
Production, quality, inventory, and finance teams often maintain separate records. This leads to data inconsistencies, reconciliation delays, and documentation gaps.
Limited Batch Traceability
Batch records, test reports, and dispatch documents are stored in different locations. Creating a complete batch history becomes time-consuming and risky during audits or recalls.
High Risk of Documentation Errors
Manual data entry increases the chances of incorrect batch numbers, quantities, and expiry dates, leading to labeling errors, delayed releases, and pharma related compliance issues.
Inefficient Deviation and Change Management
Tracking deviations, change controls, and approvals manually weakens version control and regulatory oversight.
Limited Operational Visibility
Without centralized systems, management lacks real-time insight into production status, inventory levels, and quality performance. Decisions are often based on delayed reports.
Scalability and Compliance Constraints
As volumes and regulatory obligations increase, manual systems become harder to manage, increasing dependency on individuals and operational risk. From R&D trials to commercial production and costing analysis, BatchMaster supports products throughout their lifecycle.
The Transition From Manual Systems to Generic ERP – Because Pharma Needed More
As product portfolios expanded, batch volumes increased, and compliance standards became stricter, manual systems could no longer support the growing needs of the pharma industry. They needed more support in managing batch records, quality documents, inventory, and regulatory reports.
To overcome these limitations, many pharma companies adopted generic ERP systems in an effort to digitize operations and improve basic process control. These solutions helped reduce paperwork and improve data storage, but they were not designed specifically for pharmaceutical manufacturing. This software addressed part of the problems, but some challenges were left untouched.
Limited Support by Generic ERP Systems for Process Manufacturing
Generic ERP systems cater basic operational features. They lack strong capabilities for managing formulations, potency calculations, yield variations, and intermediate processing, forcing manufacturers to rely on manual adjustments and external tools.
Weak Batch Genealogy and Traceability
Although generic ERP systems offer basic lot tracking, they do not connect process data, quality results, and compliance records into a complete batch history. This makes audits, recalls, and regulatory inspections more difficult and riskier.
Limited Integration of Quality Management
Key quality activities such as sampling, testing, batch release, deviation handling, and CAPA are often managed outside the core ERP system. This results in duplicate data entry, manual intervention, and inconsistencies.
Gaps in Regulatory Compliance Support
Pharma regulations require electronic signatures, audit trails, controlled access, and system validation. Generic ERP platforms usually need heavy customization to meet standards like GMP and 21 CFR Part 11, increasing cost, complexity, and compliance risk.
High Dependence on Customization and Add-Ons
To fit pharma workflows, companies often rely on extensive customization and third-party tools. Over time, this makes systems harder to upgrade and maintain, increasing operational risk and vendor dependency.
Lack of AI, BI, and Predictive Capabilities
Most generic ERPs lack built-in artificial intelligence, business intelligence, and predictive analytics. As a result, manufacturers miss early warnings on deviations, quality risks, demand trends, and equipment performance.
Limited Support for Regulatory and Market Expansion
When entering new markets, manufacturers must meet different regulatory requirements. Generic ERP systems lack built-in flexibility for this, requiring further customization that delays expansion and raises costs.
What Is a Purpose-Built Pharma ERP Software?
A purpose-built Pharma ERP is a specialized enterprise resource planning system designed exclusively for the unique needs of pharmaceutical manufacturing in Philippines and Southeast Asia region. Unlike manual systems that lack control and scalability, and generic ERP platforms that lack industry-specific intelligence, an ERP software for pharmaceutical industry is built to manage complex formulations, strict quality standards, and evolving regulatory requirements from the ground up.
These solutions integrate formulation management, batch processing, quality control, regulatory compliance, and end-to-end traceability into a single, validated platform. By embedding pharma-specific workflows and controls, they ensure accuracy, consistency, and compliance across every stage of production.
A purpose-built Pharma ERP enables manufacturers to operate with greater confidence, efficiency, and transparency. It not only supports day-to-day operations but also strengthens regulatory readiness, drives digital transformation, and enables sustainable business growth in highly regulated markets.
Why a Purpose-Built Pharmaceutical ERP Software Is the Need of the Hour ?
Accelerates Innovation
Connects R&D, quality, and production data with AI-driven insights to speed up product development and launches.
Enables Scalable Growth
Manages higher volumes, multiple sites, and partners using intelligent, standardized workflows.
Drives Digital Transformation
Supports paperless operations with electronic records, digital approvals, and smart automation.
Ensures Regulatory Readiness
Uses built-in compliance intelligence and audit-ready systems to adapt to changing GMP and global regulations.
Improves Business Agility
Delivers real-time dashboards and BI analytics for faster, data-driven decisions.
Reduces Manual Dependency
Replaces spreadsheets with automated controls and AI-supported validation.
Strengthens Data Integrity
Maintains accurate, secure, and traceable records with intelligent monitoring.
Supports Market Expansion
Simplifies multi-location and multi-regulatory operations with centralized intelligence.
Optimizes Costs and Resources
Uses AI and BI to analyze batch costs, wastage, and performance for higher profitability.
Why BatchMaster Pharma ERP Is Best Suited for Pharmaceutical Manufacturing?
Pharmaceutical manufacturing is a process-driven industry built on precision, documentation, and continuous quality control. Every product follows approved formulas, batch records, and regulated workflows, where even small deviations can impact compliance, safety, and business continuity.
With over 40+ years of experience, BatchMaster has worked closely with pharmaceutical and nutraceuticals manufacturers worldwide. This deep industry exposure has enabled it to understand real operational challenges, from formulation complexity and batch variability to regulatory pressure and audit readiness. Based on these insights, BatchMaster has developed a unified ERP platform that supports formulation, production, quality, compliance, inventory, costing, and reporting in one integrated solution.
Rather than offering disconnected tools, BatchMaster delivers a purpose-built platform designed to manage the complete pharmaceutical manufacturing lifecycle.

Built for Process-Based and Batch-Driven Manufacturing
Pharmaceutical production in Philippines relies on controlled formulas, bills of materials, and batch processes. Products move through multiple stages such as blending, granulation, drying, compression, coating, filling, and packaging, each with defined quality limits.
BatchMaster ERP solutions Philippines is designed for this environment. It supports advanced formulation management, controlled batch scaling, and potency-based calculations, ensuring consistent dosage, yield accuracy, and reproducibility without manual intervention.
Lot Precision and Formula Control
Raw materials often vary in potency and purity, requiring frequent adjustments. BatchMaster manages these variations through built-in potency management and automated calculations.
Formulas, bills of materials, and processing instructions are centrally controlled, helping manufacturers maintain product integrity while reducing dependency on spreadsheets.
End-to-End Traceability and Regulatory Compliance
Pharma manufacturers must maintain complete traceability from raw materials to finished products. BatchMaster provides full lot-to-batch and batch-to-lot traceability, linking process data, quality results, and compliance records into complete batch histories.
With electronic batch records, audit trails, electronic signatures, and SOP-based workflows, the system supports FDA 21 CFR Part 11, cGMP, FSMA, and global regulatory standards.
Integrated Quality Management
Quality is embedded into every stage of manufacturing. BatchMaster captures in-process testing, COA generation, deviation reporting, stability data, and CAPA workflows within the core system.
This eliminates separate quality tools, reduces duplicate data entry, and ensures only compliant batches are released.
Inventory, Expiry, and Warehouse Control
BatchMaster tracks inventory by lot, potency, and expiry date using FEFO logic. Integration with barcode scanners, mobile WMS, and electronic scales improves accuracy, minimizes waste, and prevents production delays.
Real-Time Production and Shop-Floor Visibility
Real-time visibility into batch status, resource utilization, and shop-floor activities helps manufacturers respond quickly to issues. MES-style execution tools capture weigh sheets, SOP steps, and batch progress electronically, ensuring consistent execution and reduced manual errors.
Complete Product Lifecycle Management
From R&D trials to commercial production, BatchMaster supports every stage of the product lifecycle. Built-in costing and performance tools help evaluate product viability and improve decision-making.
Flexible Integration and Deployment
BatchMaster integrates with QuickBooks, Tally, SAP Business One and Sage. It can be deployed on-cloud or on-premise, allowing manufacturers to enhance existing systems with pharma-specific intelligence.
Support for Nutraceutical and Allied Segments
BatchMaster includes nutraceutical capabilities such as potency-based formulation accuracy, label-claim management, allergen tracking, and digital batch records, enabling multiple regulated product lines to be managed on one platform.
Audit-Ready Documentation and Reporting
Centralized batch tickets, audit trails, SOPs, stability data, and deviation logs ensure fast access to validated documentation. This simplifies inspections and reduces audit risk.
Conclusion: Enabling Compliance, Quality, and Growth
BatchMaster Pharma ERP brings together formulation control, integrated quality management, regulatory compliance, and operational visibility in one purpose-built solution, powered by advanced AI and BI capabilities for smarter, faster decision-making. With intelligent analytics, predictive insights, and real-time dashboards, BatchMaster Web ERP enables manufacturers to identify risks early, optimize processes, and improve overall performance. Its built-in AskBME assistant provides guidance on how to perform tasks using simple prompts, enhancing productivity.
For pharmaceutical manufacturers seeking to reduce risk, improve efficiency, and scale confidently in regulated markets, this pharmaceutical ERP delivers a proven, industry-focused platform for long-term success.
To learn how BatchMaster can transform your pharmaceutical operations, connect with our experts today and explore a solution built specifically for your industry. Request a demo Today.




